Hawkinson also serves as Silverback's Senior Vice President of Finance and principal accounting officer. Odegard as of the Transition Date will continue to vest and will remain exercisable during the consulting period.Įffective as of the Transition Date, Russ Hawkinson has been appointed to serve as Silverback's principal financial officer. Odegard will be paid an hourly rate of $350 and all outstanding equity awards held by Dr. As consideration for each of their consulting services, Dr. Odegard and have each entered into a consulting agreement with Silverback as of the Transition Date pursuant to which each has agreed to provide, on an as-needed basis, not to exceed 20 hours per week unless mutually agreed, transition services and to advise, consult and support management team in connection with the closing of the potential merger of a wholly owned subsidiary of the Company with and into ARS Pharmaceuticals, Inc., a Delaware corporation (the Merger), winddown activities related thereto, the sale of legacy assets and other services from the Transition Date until the later of (a) the closing of the Merger and (b) November 30, 2022. Odegard and to pursue other employment opportunities. approved the termination of employment of Valerie Odegard, Ph.D., President and Chief Scientific Officer, effective as of Septem(the Transition Date), to extend cash runway and to allow Dr. Initially the deal’s size was 7 million shares at $17 to $19.On September 1, 2022, the board of directors of Silverback Therapeutics, Inc. (Silverback Therapeutics increased its IPO at pricing: 11.5 million shares at $21, up from 10 million shares at $19 to $20, which were the larger terms in its SEC filing on Dec. Our approach is designed to increase the therapeutic window and avert unacceptable toxicities by directly targeting specific disease sites where our therapeutics are locally active. 
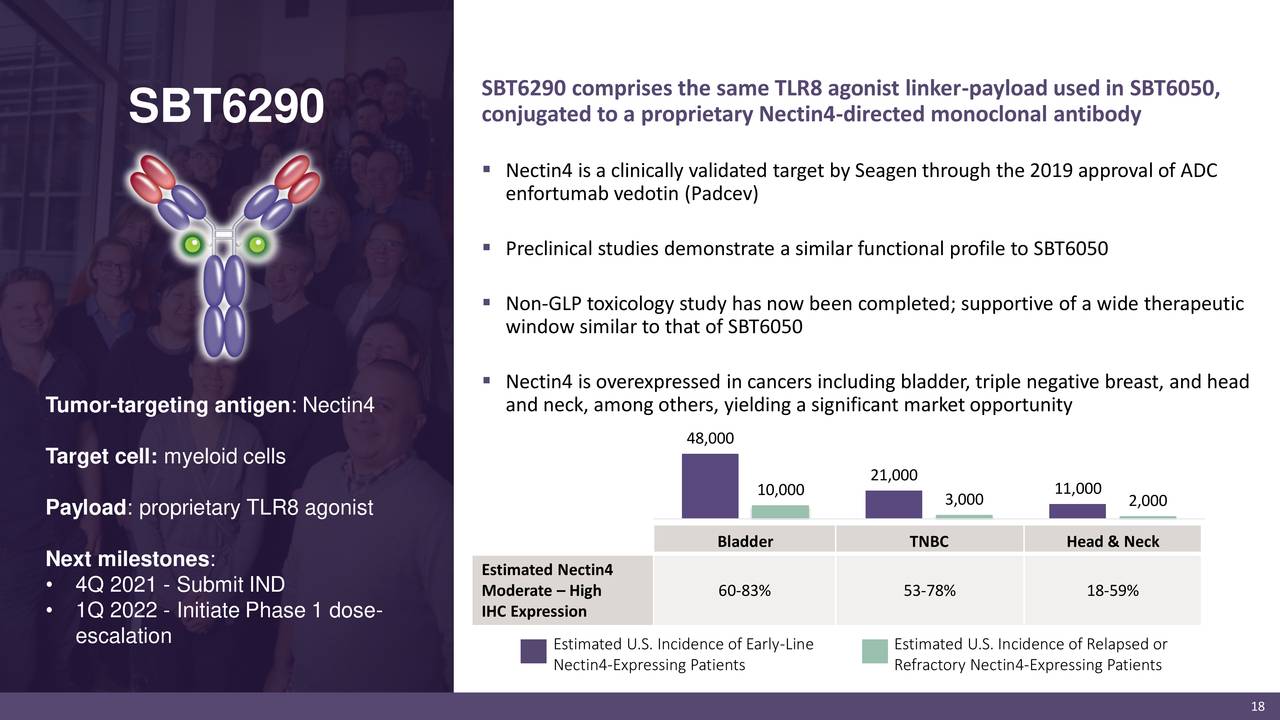
Many potentially promising systemic therapies fail to maximize their therapeutic potential due to toxicities in healthy tissues. Our ImmunoTAC platform is the result of a focused effort to discover ways to systemically deliver disease-modifying small molecules in a directed fashion to sites of disease. Our third TLR8 program, SBT8230, is comprised of a TLR8 linker-payload conjugated to an ASGR1 monoclonal antibody that is under development for the treatment of chronic hepatitis B virus infection (cHBV). We anticipate submitting an investigational new drug application (IND) for SBT6290 in the fourth quarter of 2021. SBT6290 is a TLR8 linker-payload conjugated to a monoclonal antibody that targets Nectin4, which is expressed in certain bladder, triple negative breast, head and neck, and non-small cell lung cancers. 
SBT6290 is our second product candidate, expanding on the potential of a TLR8 agonist as a payload. In this trial, we have observed changes in pharmacodynamic markers in the first dose cohort, and we anticipate providing an update on interim data from the Phase 1 dose-escalation cohorts in the second half of 2021. SBT6050 is currently in a Phase 1/1b clinical trial in patients with advanced or metastatic HER2-expressing solid tumors. 
Our lead product candidate, SBT6050, is comprised of a TLR8 agonist linker-payload conjugated to a HER2-directed monoclonal antibody that targets tumors such as certain breast, gastric and non-small cell lung cancers. Initially, we are applying our platform to create a new class of targeted immuno-oncology agents that direct a myeloid cell activator to the tumor microenvironment (TME) in solid tumors to promote cancer cell killing. We are a clinical-stage biopharmaceutical company with one product candidate in a Phase 1/1b clinical trial, and we are focused on leveraging our proprietary ImmunoTAC technology platform to develop systemically delivered, tissue targeted therapeutics for the treatment of cancer, chronic viral infections, and other serious diseases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
